This is probably the moment you’ve been waiting for. We’re about to uncover the mystery of everyone’s favorite science experiment: Mentos and soda.
It’s like middle school all over again, isn’t it? We know we can’t be the only ones who begged our parents to let us conduct this experiment in the backyard. And let’s face it— there are few things more interesting than the aftereffects of mixing Mentos with a carbonated beverage.
In case you have no idea what we’re talking about, putting Mentos mints in a fizzy drink like Coke creates an explosion. That’s right! The reaction causes the beverage to spurt out of the container and make its way into the air. If you think about it, it’s like a soda fountain.
Now we know what you’re thinking: why do Mentos react the way they do? What is it about soda that leads to a giant eruption? Well, lucky for you, the truth will finally be revealed. Keep reading for answers to every single one of your questions!
Why Do Mentos React With Coca-Cola?
Everyone knows carbonated beverages contain carbon dioxide. What most people don’t know is that the surface of a Mentos mint is full of craters, which means it’s the perfect site for bubbles to form. So naturally, as the bubbles accumulate, the liquid will be forced to release and erupt. Boom!
Alright, it’s showtime. We’re about to dive into the nitty-gritty of what might be the most astonishing experiment of all time. So, are you ready for possibly the best science lesson of your life?
What Goes On Inside Soda
Let’s start from the beginning, shall we? Soda is full of a little something called carbon dioxide gas. This gas forms a bond with water and ultimately creates the fizzy drink you know, love, and devour with all your heart.
Soft drink manufacturers pressurize the beverage to keep it as fizzy and pleasant as possible for you. When you open a bottle or pour some soda into a glass, you’ve probably noticed a handful of bubbles rise to the surface. Why does this happen?
The truth is that the gas bubbles are dying to escape. Seriously. As you gently twist the bottle to consume the drink, you equalize the pressure and set some bubbles free.
Though releasing the pressure forces some of the little gas bubbles out of solution, most of them remain in the same place due to the surface tension of the water. And to put it simply, they’re desperate to escape.
Now here’s the thing. To escape, the carbon dioxide molecules search left and right for a rough surface. This surface will allow the molecules to gather together in a process named nucleation, which leads to the formation of bubbles.
How Mentos Add to the Equation
Enter Mentos. Believe it or not, these mints have thousands of microscopic pores and peaks. That’s right! Don’t fall for the outward appearance. As the old saying goes, looks can be deceiving.
So, in short, what we’ve got here is a large, rough surface. And the microscopic craters we just mentioned serve as nucleation sites, which are places for the bubbles to grow.
The role of the surface is to break the bond between carbon dioxide gas and water and create the iconic eruption that we’re all dying to see. You know the one.
Mentos are more potent than we give them credit for, you know. When dropped in soda, they act as surfactants. And what are those, you ask? Surfactants reduce the surface tension between two liquids or a liquid and a solid. Pretty cool.
In simple words, they catalyze the process that makes fizzy drinks fizzy. As you already know, gently releasing the pressure causes little gas bubbles to rise and allows you to enjoy a delicious carbonated beverage. So similarly, adding a larger surfactant like Mentos causes more bubbles to come to the surface, leading to a massive explosion. It’s the same thing but on a bigger scale.
It’s important to remember that multiple factors can affect the eruption’s size. The speed of the Mentos, for instance, can make a difference. So can the temperature, viscosity, and chemical makeup of the liquid! Every detail matters.
Technically, you can conduct this experiment with just about any carbonated beverage. The fizz makes the magic, after all. But as you’ve probably noticed, the most popular soda option tends to be Coke. And honestly, we can see why.
Do Mentos React Better With Regular Coke or Diet Coke?
Fun fact: Mentos react better with Diet Coke than regular Coke! Who would’ve thought? You see, Diet Coke contains artificial sweeteners like aspartame, which lower the surface tension even more. As a result, the soda explosions are bigger and better!
We bet you didn’t see that one coming, did you? It’s true! Diet Coke and Mentos are a match made in heaven. Here’s why.
As you know, Diet Coke doesn’t contain any sugar, unlike classic Coke. But to make up for the lack of sugar, the company uses artificial sweeteners as sweetening agents.
According to Coca-Cola, here’s what you’ll find in a bottle of Diet Coke:

- Carbonated water
- Caramel color
- Aspartame
- Phosphoric acid
- Potassium benzoate
- Natural flavors
- Citric acid
- Caffeine
Do you spy a little something named aspartame? That right there is an artificial sweetener that is 200x sweeter than regular sugar. Yes, you heard that right!
Let’s not overlook all the other additives in the drink, too. Potassium benzoate, for instance, is a food preservative that restricts the growth of bacteria. And citric acid is a common flavoring and preserving agent.
Why does this matter, you wonder? According to a study by Spring Arbor University, certain beverage compounds affect the soda fountain’s overall height.
Here are the effects of the solutes:
- Aspartame creates a fountain up to 3x the original height
- Benzoate creates a fountain up to 3x the original height
- Citral creates a fountain up to 4x the original height
- Linalool creates a fountain up to 4x the original height
- Citric acid creates a fountain up to 6x the original height
And as you know, Diet Coke contains aspartame, potassium benzoate, and citric acid. Do you see where we’re going with this?
Here, have a look at the results yourself:
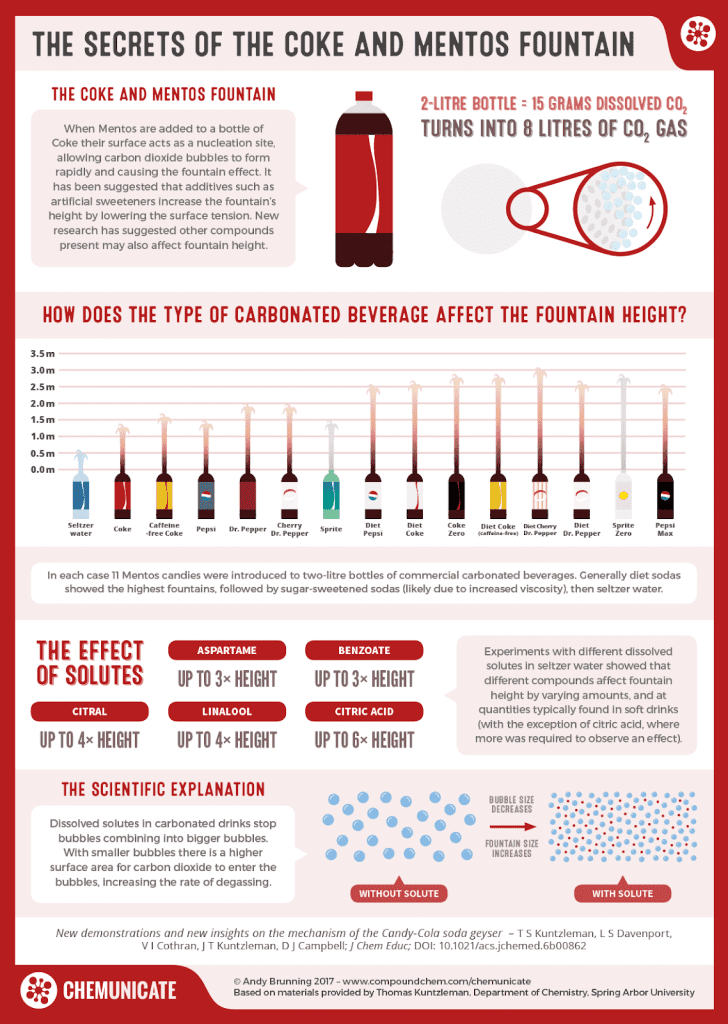
As you can see, diet sodas like Diet Coke led to the highest fountains. And it makes perfect sense, too! Artificial sweeteners like aspartame lower surface tension, which naturally increases fountain height. Combine those with other additives, and you’ve got an eruption to remember!
Isn’t science just the most remarkable thing? So yes, if you decide to have fun with Mentos and soda, grab a Diet Coke if you want to see some action. It’ll make all the difference in the world.
How Many Mentos Do You Put in a Coke to Make It Explode?

The exact number of Mentos is just a personal preference! We advise putting in more than one to get things going. Consider buying a roll and playing around with the amount for some fun! A handful of mints should do the trick.
Will Mentos Work With Flat Coke?
The answer is simple: no. Flat Coke is Coke that isn’t as fizzy, which means that the dissolved carbon dioxide has officially bubbled out. And as you know, carbonation is significant to the Mentos-Coke experiment! So no, flat soda will not give you the results you want.
Does It Have the Same Reaction in the Body?
Contrary to popular belief, Mentos and Coke will not cause an explosion in your body. In fact, it’ll have little to no effect. You might experience mild discomfort, but it won’t do much harm. Feel free to browse the depths of YouTube for proof!
Conclusion
Honestly, we feel like we’re in middle school again. And honestly, we kind of love it.
There’s something so exhilarating about watching a soda geyser before your own eyes. It’s the sort of adrenaline rush that makes us feel like we’re alive, you know?
If you want to try it out, buy a Diet Coke and a roll of Mentos! Grab your safety goggles while you’re at it, and be sure to perform this experiment in an open space. It can get a little messy, after all.
The key is dropping the Mentos as fast as possible and running away before things get explosive. Oh, and don’t forget to record a video to capture the epic reaction forever! This is one memory you won’t want to forget.








